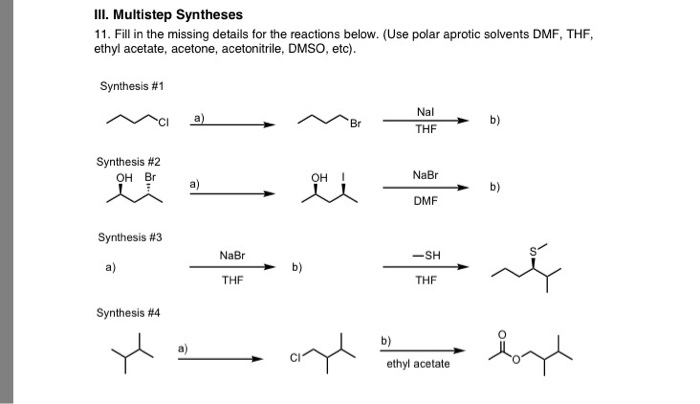
Isobaric Vapor–Liquid Equilibria of Binary Mixtures of γ-Valerolactone + Acetone and Ethyl Acetate | Journal of Chemical & Engineering Data
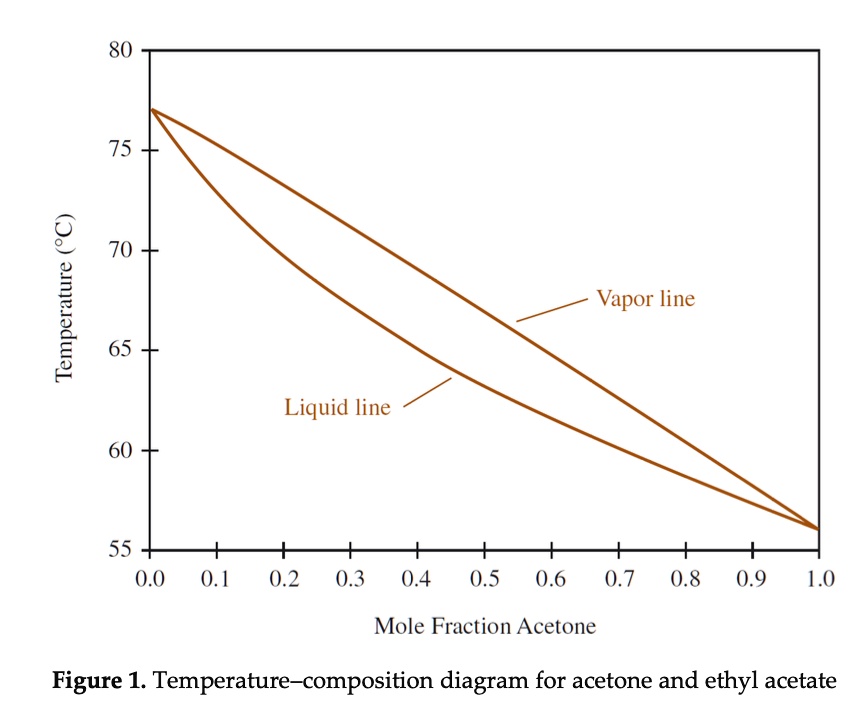
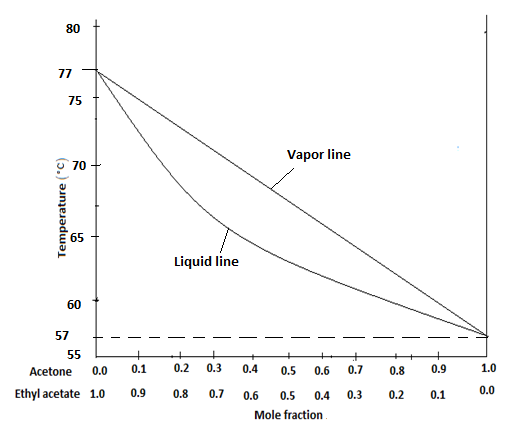
SOLVED: 80 75 70 Temperature 65 Vapor line Liquid line 60 55 0.0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1.0 Mole Fraction Acetone Figure 1. Temperature-composition diagram for acetone and ethyl acetate
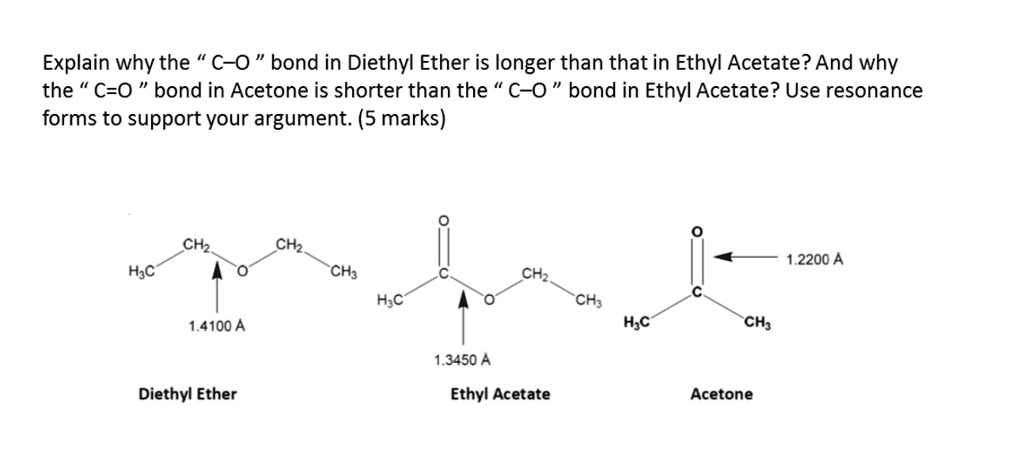
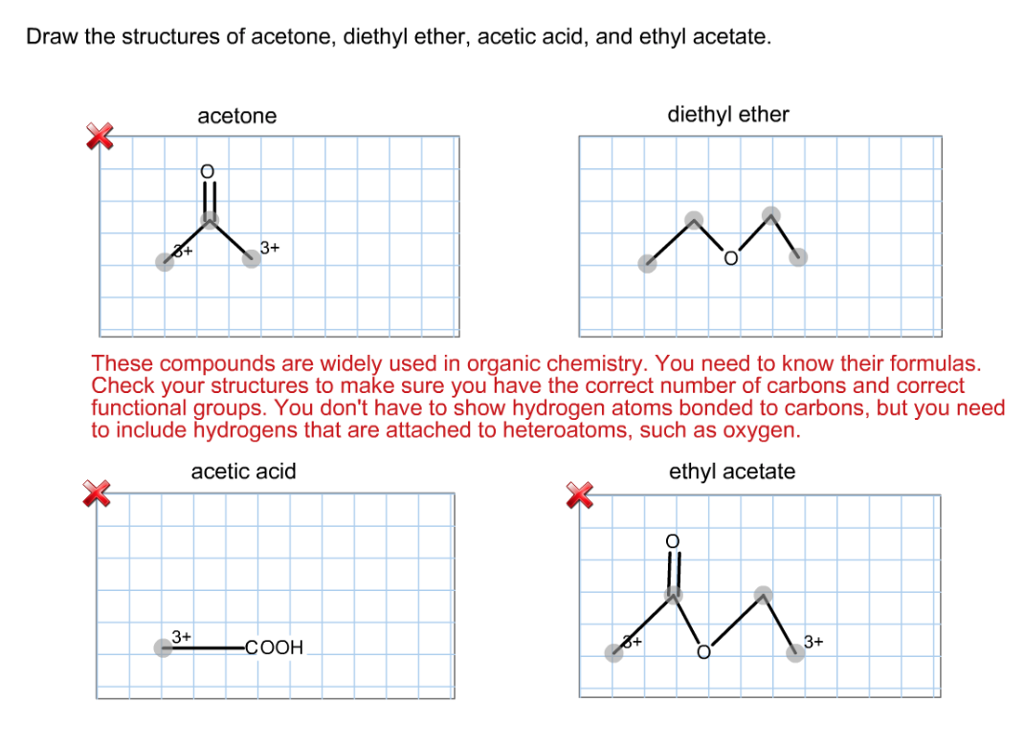
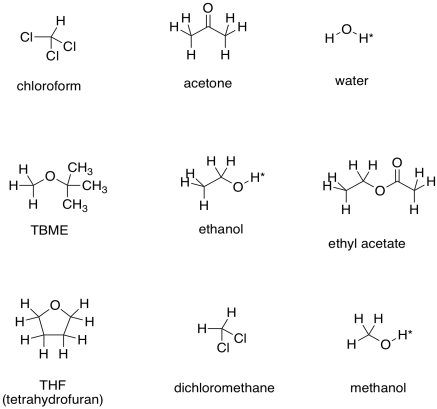
SOLVED: Explain why the C-O bond in Diethyl Ether is longer than that in Ethyl Acetate? And why the C=O bond in Acetone is shorter than the C-O bond in Ethyl Acetate?